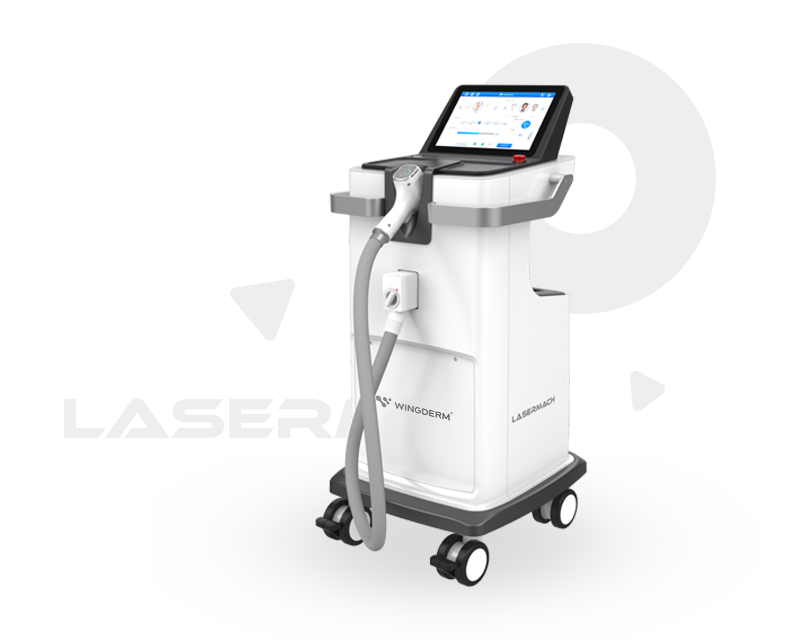
Wingderm® since its establishment in 2016, with the aim of "Aesthetics & Technology, Easy to Achieve" , provide leading and reliable intelligent medical aesthetic devicesfor beauty seekers worldwide,introducing more people to the aesthetic experience brought by technological innovation.
With the technology innovation, Wingderm® has successively released multiple intelligent medical aesthetic devices, which have been exported to more than 80 countries, including those in Europe, America, and the Middle East, with over 25,000 units installed, recognized for safety and effectiveness by both experts and beauty seekers.